In pickling of steel, to remove rust or iron oxide scale from iron or steel before subsequent processing, such as extrusion, rolling, galvanizing, and other techniques. Technical quality HCl at typically 15 to 18% concentration is the most commonly used pickling agent for the pickling of carbon steel grades.
Pickling is the chemical removal of surface oxides (scale) and other contaminants such as dirt from metal by immersion in aqueous acid solution. Acid reacts with the oxides and base metal to produce dissolved metal salts, thereby neutralizing the acid.
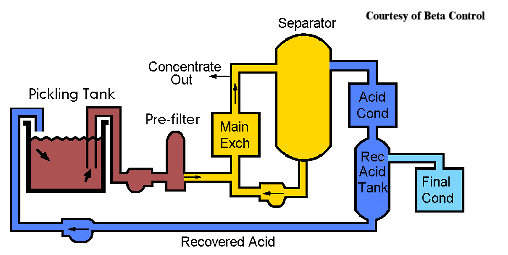
The pickling process generates considerable quantity of spent pickle liquor (SPL) containing the dissolved metal salts as well as residual free acid. This SPL must be disposed of by chemical neutralization with alkali which consumes large chemical requirement or regenerated which is the safest way of economics and environment care.
Although carbon steel can be pickled in either sulphuric or hydrochloric acid, for quite some time hydrochloric has been the acid of choice, particularly for large, continuous, strip lines.
Hydrochloric acid offers a number of potential advantages over sulphuric acid, including the following:
• It consistently produces a uniform light gray surface on high-carbon steel
• Less chance of over-pickling
• Iron concentrations can be as high as 13%
• Rinsing is facilitated because of the high solubility of iron chloride
• Safer to handle • Lower operating temperatures can be employed
Hydrochloric acid regeneration refers to a chemical process for the reclamation of bound and unbound HCl from metal chloride solutions such as ferrous chloride. Regenerated acid has no adverse effect on metal cleaning efficiency compared to virgin acid. It pickles as efficiently as virgin acid. |